The US Food and Drug Administration (FDA) has cleared the initiation of a pivotal clinical trial evaluating LensOne, an investigational prosthetic capsular bag developed by Long Bridge Medical for secondary intraocular lens (IOL) fixation in eyes lacking adequate capsular support.
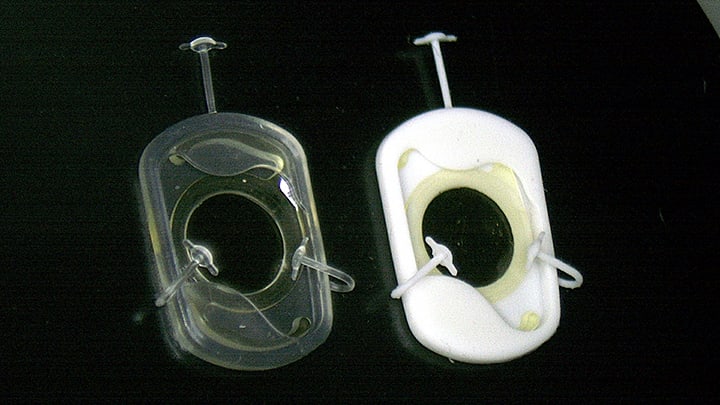
The FDA’s investigational device exemption (IDE) approval allows the launch of the GOLDEN GATE trial, a prospective, multicenter study assessing the safety and effectiveness of LensOne in patients with IOL dislocation who are poor candidates for anterior chamber IOLs. The device previously received the FDA’s breakthrough device designation, reflecting a lack of approved surgical options for this patient population.
Secondary IOL fixation remains a challenge in eyes with insufficient capsular support, often requiring technically complex approaches such as scleral suturing or intrascleral haptic fixation. These methods are associated with variability in outcomes and complication risk, and no standardized solution is widely adopted.
LensOne is designed to recreate a capsular bag–like structure, enabling placement of a posterior chamber IOL using familiar surgical techniques. In a first-in-human study of 15 patients, 12-month results suggested a favorable safety and performance profile, supporting further evaluation in a US pivotal trial.1
Enrollment in the GOLDEN GATE study is expected to provide data on reproducibility, safety, and clinical outcomes to inform potential regulatory approval. RP
Reference
1. Brodie F. Implantable artificial lens capsule for secondary IOL—first in human results. Presented at: 2025 Vail Vitrectomy Meeting; February 11, 2025; Vail, Colorado.








